Fanconi anemia proteins : guardians of the genome
First Prize in the 2026 Blog Contest, by Mélissa Thomas.
April 23, 2026
Anemia. You’ve likely heard that word before, indicating a lack of red blood cells, the principal component of blood. “Fanconi”, though ? You’re probably not familiar with that Italian-sounding name, but that’s understandable. Discovered in the 20s by Dr Fanconi, a pediatrician actually not from Italy but from Switzerland, Fanconi anemia is a rare disease, occurring in 1 in 130 000 individuals worldwide. As the name suggests, one of the major symptoms of the disease is anemia, but that’s unfortunately not all. Patients are usually born with malformations, such as an extra finger or a small thumb, and are up to 800 times more likely to develop cancer (1) (2). You may now be wondering : how can the same disease cause anemia, finger anomalies and cancer ? To answer that question, we need to look a little deeper, at the Fanconi anemia proteins.
The FANC proteins : 23 friends for life
“Fanconi anemia proteins” is a bit of a mouthful, so we’ll use their shorter name from now on : FANC proteins. Before we go any further, let’s make sure we’re on the same page when it comes to proteins.
While DNA is like a manual, whose instructions can be read over and over again, proteins are the effectors of these instructions. No offense to DNA, but proteins are the ones actually doing the hard work. They are builders, communicators, recyclers, repairers, energy engineers, transporters, fighters and much more. Anything the body needs to function, proteins do it.
There are 23 FANC proteins, all working together in the same pathway to repair a specific type of damage that can occur in cells : crosslinks. A mutation, an error, in just one of these 23 proteins, is what causes the Fanconi anemia disease.
Crosslinks and how to repair them
Our cells regularly encounter chemical compounds called crosslinkers. One of the major crosslinkers found around us is formaldehyde (3). It’s hiding in resins and paints, in smoke from cigarettes and combustion, in alcohol and in many food items such as shiitake mushrooms, crustaceans and pears. More problematically, formaldehyde also comes from within us : we can’t escape it. It’s a by-product of many every-day processes happening in cells, such as the ones that regulate which parts of the DNA, the manual, should be read.
Formaldehyde is a tiny compound, 375 000 times smaller than a hair strand, so it can very easily sneak around the cell and get to the DNA. What does it do there ? Crosslinkers are like gum in a way, they can interact with the DNA area they reach and make it sticky, creating a knot with another nearby DNA area. To get back to my manual metaphor : picture a piece of gum stuck between 2 pages – that’s a crosslink ! As you can imagine, this crosslink is making reading the manual difficult. And that’s what happens : these areas of DNA are made inaccessible, and their instructions cannot be read.
Not to worry though, we have proteins in our cells that can remove this formaldehyde, called ADH5 and ALDH2. However, just like everything in life, they’re not perfect, and a small amount of formaldehyde escapes them and does reach DNA, creating crosslinks. That’s when FANC proteins come into play.
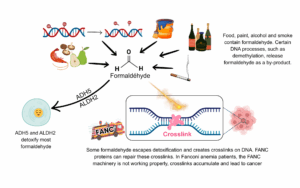
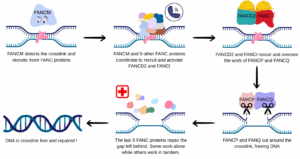
FANC proteins can detect crosslinks and remove them, by cutting around the “knotted DNA” and repairing the hole left behind. Each FANC protein plays a different role in this cascade of event : FANCM is in charge of detecting the crosslink, FANCD2 and FANCI are coordinating the cutting step, itself done by FANCP and FANCQ, and FANCD1 and others are performing the delicate step of faithfully restoring the removed DNA (4).
This ensures that no matter the number of pears and mushrooms you eat, your DNA stays crosslink-free and readable !
Fanconi anemia disease
In Fanconi anemia patients, this FANC machinery is not working properly. Crosslinks accumulate on DNA, the manual can’t be ready properly and errors in how the cells function arise : errors during embryonic development, errors in red blood cell production and errors in cell growth control, leading to malformations, anemia and cancer.
Anemia is now well-managed through bone marrow transplants, but cancer is still a major challenge for Fanconi anemia patients who cannot be treated by conventional anti-cancer therapies. In our lab, we’re working on FANC proteins and their partners, in order to better understand how these Fanconi anemia cancers emerge and the role of formaldehyde in driving them (5) (6).

Mélissa Thomas
Mélissa is a postdoctoral fellow at the Cancer Research Center in Québec, whose work focuses on DNA repair in the context of oncology. She’s a science communicator, passionate about bringing science to everyone, particularly through Soapbox Science which she co-organizes in Québec.
References:
- For more information on Fanconi anemia, see the Fanconi Anemia Cancer Foundation website https://fanconi.org/.
- Pour de l’information en français sur l’anémie de Fanconi, voir le site internet de l’Association Française de la Maladie de Fanconi https://www.fanconi.com/
- A publication on the toxic effect of aldehydes. Vijayraghavan, Sriram, and Natalie Saini. “Aldehyde-associated mutagenesis─ current state of knowledge.” Chemical research in toxicology 36.7 (2023): 983-1001.
- A thorough but short review on the Fanconi anemia pathway. Rodríguez, Alfredo, and Alan D’Andrea. “Fanconi anemia pathway.” Current Biology 27.18 (2017): R986-R988.
- Some of our lab’s work on Fanconi anemia. Dubois, Emilie L., et al. “A Fanci knockout mouse model reveals common and distinct functions for FANCI and FANCD2.” Nucleic acids research 47.14 (2019): 7532-7547.
- Our latest published work on Fanconi anemia. Gao, Yuandi, et al. “A CRISPR-Cas9 screen identifies EXO1 as a formaldehyde resistance gene.” Nature communications 14.1 (2023): 381.




















